Why Does Hot Air Go Up?
And Why Do I Care?
For my explanation I have created a model home simulating 32 feet of elevation. The inside ceiling is flat. The pitched roof just makes it look like a house and makes room for the numbers.
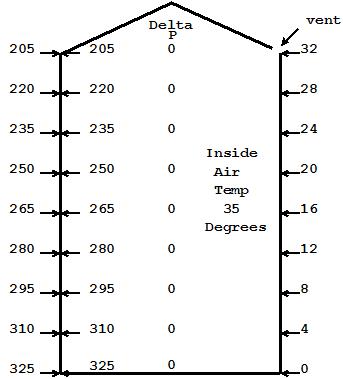
We start with Fig 1 where our outside and inside air temperatures are both 35 degrees. There is a vent at the top, so both outside and inside air pressures are the same and thus the delta P at all levels is zero and no air would move in or out if we provided a path.
The 325 at the bottom of our house represents 101,325 Pascals of atmospheric pressure, the same as the more common reference of 14.7 psi at sea level. For convenience I have dropped the 101, but it is still there.
The pressure drop per foot of elevation as you go up is 3.75 Pascals, or 15 Pascals for every 4 feet. see Note 1 below.
Fig 1
Outside
Air
Temp
35
Degrees
Outside
Air
Temp
35
Degrees
Outside
Air
Temp
35
Degrees
101,325
Pascals
101,205
Pascals
3.75
Pascals
for every
foot
of
elevation
Note 1: At sea level, the atmospheric pressure is 14.7 psi. At 10,000 feet the pressure is 10.1 psi or 31.29% less. If we convert that to Pascals, our 101,325 Pascals has been reduced by 31705 Pascals. Divide that number by 10,000 feet and we see a drop of 3.17 Pascals for every foot we go up. Since that is the average over the 10,000 feet we can adjust the lower region to 3.75 Pascals per foot to be more accurate. If you live in Denver, just use 3.
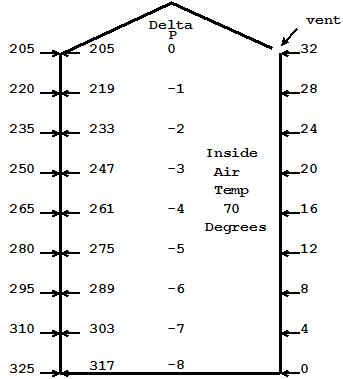
Fig 2
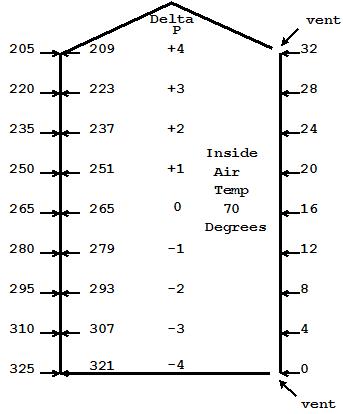
Fig 3
Note 2: The weight of a given volume of air is inversely proportional to its (absolute) temperature. Double its temperature and you halve its weight per unit of volume. However, the temperature has to be converted to Kelvin. So our increase from 35 degrees F to 70 degrees F is actually an increase from 275 K to 294 K. Dividing that increase of 19 degrees K by 294, I get 6.5% representing the volume of air forced out during expansion. Thus an increase of 35 degrees F will give us a 6.5% decrease in weight. Where our 35 degree outside air decreases at 3.75 pa per foot of increased elevation, our warm air inside decreases at 6.5% less or about 0.25 Pascals per foot. Conveniently, that is 1 Pascal per 4 feet as illustrated in our diagram.
For the most part we could continue to believe that hot air has some magical power to move up when heated, but besides that being wrong, every time someone explains it that way or even implies it, we take another step backwards. And if you don't think those little steps count, just look at the number of people who have followed that path in the wrong direction and now talk about warm air rising all by itself. So, from now on, even when it doesn't seem to matter, we should establish talking points that will allow us to talk about air flow, the warm and the cold, and yet not imply that warm air will go up all by itself. Those talking points are yet to be discussed or determined as this is just the beginning. To start, I want to describe stack effect, a form of air movement important to our energy profession and one that we should all fully understand.
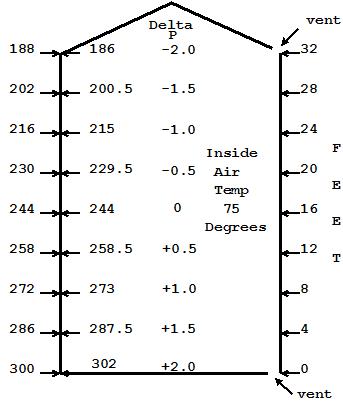
Fig 3, I have added a vent at the bottom. With the 8 pa pressure difference at the bottom from Fig 2, air begins to flow in, increasing the pressure from bottom to top within our home. If the pressure at the bottom increases 1 Pascal, then the pressure at the top will increase the same, as the weight of the column has remained the same. We are assuming that the incoming air is being heated to 70 degrees as it enters.
As the pressure inside the house increases, the delta P at the bottom decreases and the delta P at the top increases. As the air flow continues, we eventually reach a balance where the air exiting the top equals the air coming in the bottom. If we consider our upper vent to be a bit larger than the bottom vent, then the expansion of the incoming air as it was heated as it came in will have been accounted for and our neutral plane will be right in the middle where shown.
In Fig 2, I have increased the inside air temperature to 70 degrees. Since the ceiling is vented, the expanded air simply escaped the top and the inside to outside air pressure at the top equalized at 205, thus the air flow stopped. Since the warm air inside is lighter than the outside, see Note 2, I then added the weight of each foot of warm air (now 3.5 Pascals per foot) on the inside to the 205 as I moved down in the air column inside the house. 70 degree air is approximately 0.25 pa lighter per foot than 35 degree air, so for every 4 feet I moved down I accumulate 1.0 Pascal of delta P between inside and outside. Over the 32 feet, the total pressure difference from top to bottom inside our house is 8 Pascals less than on the outside.
However, this is not what the pressure distribution in our homes looks like. As we have seen and have been taught, the cold air enters the bottom and the warm air exits the top, and somewhere in the middle there is a neutral plane where inside and outside pressures are the same.
Welcome
to
"My" Energy Workshop
Hot - Air
********************************************************************************************************************************************
Reverse Stack Effect in Summer
Outside
Air
Temp
100
Degrees
Outside
Air
Temp
100
Degrees
The description for reverse stack effect is currently in progress along with several other supporting pages. Links to all will be provided here when completed.